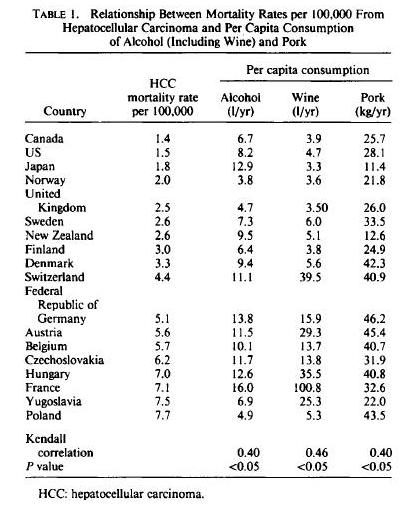
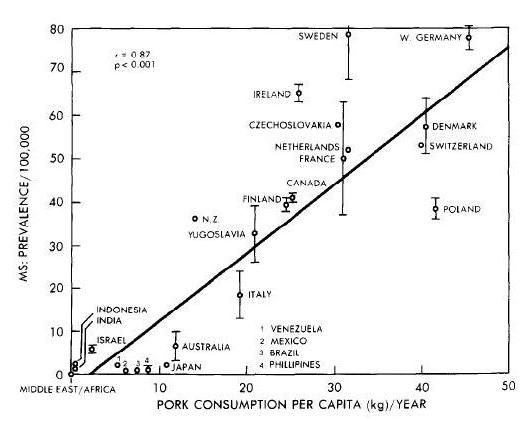
We started this series with a look at remarkably strong correlations between pork consumption and liver cirrhosis mortality, liver cancer, and multiple sclerosis (Pork: Did Leviticus 11:7 Have It Right?, Feb 8, 2012). In Part 2, we looked at omega-6 fats in industrial pork meat and toxins in processed pork products as possible causes (The Trouble with Pork, Part 2, Feb 15, 2012).
That second post left us with several clues that some pathogen (or pathogens) that (a) infects both pigs and humans and (b) can be transmitted from pigs to humans via the eating of pork, is responsible for the disease associations. These clues include:
- The risk is higher for fresh pork than processed pork. Processed pork is generally cured or smoked, both steps that are anti-microbial.
- Eating fiber, which increases gut bacterial populations and enhances immune vigilance of the gut, is protective.
- The disease risk is specifically associated with two organs – the central nervous system (multiple sclerosis) and the liver (cirrhosis, hepatocellular carcinoma). Pathogens are more likely than other pork components to have tissue specificity.
Our mission today is to try to track down the pathogen(s), and figure out how to minimize risk of infection.
Pigs And Zoonotic Infections
Scientists studying xenotransplantation – the transplantation of animal organs into a person to replace a failing organ – have had the best luck with pig organs. Pigs are easier to work with than primates, not dramatically different in size than humans, and their organs are less likely to provoke rejection than those of other mammals. This suggests a similarity of biology between pigs and humans.
But biological similarity has its downsides. A large number of pathogens can infect both pigs and humans. More than any other animal, pigs pass pathogens to humans.
Indeed, investigators have been surprised at how frequently pathogens pass back and forth. According to a new study (discussed at Aetiology) of the evolutionary history of methicillin-resistant Staphylococcus aureus (MRSA), S. aureus was passed to pigs by their human caretakers. In pigs, which are routinely given antibiotics by industrial food producers, S. aureus picked up resistance genes to tetracyclines and methicillins. The resulting antibiotic-resistant ST398 strain was passed back to humans.
Wikipedia lists some of the pathogens that flourish in both pigs and humans and can infect humans who eat infected pork, usually undercooked pork:
- Pork tapeworm, Taenia solium, not only causes a parasitic infection of the intestines (the worm is typically 2 to 3 meters long) but also the larvae can infect the brain and muscles, causing the disease Cysticercosis which is a common cause of seizures.
- The roundworm Trichinella spiralis causes trichinosis. As in tapeworm infections, the mature worms are largely confined to the intestines where symptoms include nausea, heartburn, dyspepsia, diarrhea, and eosinophilia but larvae can migrate through the body, producing edema, muscle pain, fever, and weakness. The greatest danger is if the larvae reach the brain, where they can cause “serious neurological deficits (such as ataxia or respiratory paralysis), and even death.” Death results from a rare form of stroke, myocarditis, encephalitis, or pneumonia.
- Other helminths including pinworms and hookworms, and ascarid worms. [14]
- Listeria monocytogenes [24].
- E. coli, Salmonella, and Staphylococcus aureus [25].
- Yersinia enterocolitica, which causes gastroenteritis, is nearly always transmitted through pork.[28]
- Brucella spp., the bacteria which cause brucellosis or undulant fever.
- Various viruses, including porcine herpesvirus, rotavirus, parvovirus, and circovirus. Some influenza viruses, such as the famous “swine flu,” have been known to be transmitted from pigs to humans. SARS is an example of a coronavirus which was transmitted from pigs to humans. PERVS (porcine endogenous retroviruses) may also be infectious in humans; of three subgroups of infectious PERVs (PERV-A, PERV-B, and PERV-C), PERV-A and PERV-B have been shown to infect human cells in culture. [24][26]
Although all of these pathogens are potential concerns, I do not see strong specific links between the above pathogens and our three pork-associated diseases – liver cirrhosis, liver cancer, and multiple sclerosis.
However, there is another pathogen capable of infecting humans from pork that is a strong candidate: hepatitis E virus (HEV).
Hepatitis E
Hepatitis E was first observed in a 1955 outbreak in New Delhi, India. It generally produces an acute disease that lasts for several weeks; most victims recover with few symptoms, but in a few this acute illness progresses into a severe liver disease that can be fatal. About 2% of all infections lead to death from this acute liver disease; death rates are higher in pregnant women.
Hepatitis E seems to have evolved in the last millennium: There are four known genotypes, all of which infect humans and two of which infect pigs, and their common ancestor dates to 536 to 1344 years ago. [1] However, the pig-infecting genotypes 3 and 4 of Hepatitis E underwent a notable population expansion in the twentieth century, during which there has been “an extensive genetic divergence of HEV strains and high prevalence of HEV infections in many parts of the world.” [2]
The human-only genotypes of Hepatitis E are transmitted by fecal contamination of drinking water and are prevalent only in developing countries with poor sanitation; but the pig-and-human genotypes are transmitted primarily through pork consumption:
[G]enotypes 3 and 4 are associated with sporadic disease attributable to exposure to body fluids of infected swine [8] and ingestion of food products from pigs, boars and deer [11], [16], [18]. [1]
Hepatitis E seems to be most prevalent in Asia, the Middle East, and Africa:
Hepatitis E is the most important or the second most important cause of acute clinical hepatitis in adults throughout Asia, the Middle East and Africa. [8]
However, it has been spreading to Europe and the Americas:
HEV was rarely identified in industrialized countries, and the few reported cases of infection were usually in someone who had recently traveled to an endemic region. In the past few years this pattern has changed, as cases of endemic or autochthonous hepatitis E have been diagnosed with increasing frequency in individuals who have not traveled abroad….
Cases have been reported with increasing regularity throughout Western Europe, as well as in some Eastern European countries. [7]
The genotypes that coinfect humans and pigs may have originated in East Asia:
All but one genotype 4 sequence originated either from China or Japan…. [T]he genotype 3 sequences were divided into 3.1 and 3.2 clades … [A]lthough 87.5% of the clade 3.1 variants were from Asia and 60% of the clade 3.2 variants were from Europe (Table S1), these clades were found to have similar histories (Fig. 6). [1]
Historically, China and Japan did not raise cattle for food and pigs have been the major source of meat. Even today in southern China, pigs are often kept in the yards of homes, and close contact between pigs and humans facilitates zoonotic transmission.
At pig farms, Hepatitis E virus seems to spread readily. A Japanese study reported:
[O]ur estimates imply that more than 95% of pigs are infected before the age of 150 days. [3]
Presumably this is due to fecal-oral transmission among pigs in close quarters. At French farms, 65% of pigs were found to be hepatitis E infected at age 90 days. [4]
Transmission to Humans Via Pork
Can humans get infected by eating pork products? It now seems clear that the answer is yes.
A French study found that the genotype distribution of hepatitis E infecting humans is identical to the genotype distribution in pigs at slaughterhouses:
Frequent zoonotic transmission of hepatitis E virus (HEV) has been suspected, but data supporting the animal origin of autochthonous cases are still sparse. We assessed the genetic identity of HEV strains found in humans and swine during an 18-month period in France. HEV sequences identified in patients with autochthonous hepatitis E infection (n = 106) were compared with sequences amplified from swine livers collected in slaughterhouses (n = 43). Phylogenetic analysis showed the same proportions of subtypes 3f (73.8%), 3c (13.4%), and 3e (4.7%) in human and swine populations. Furthermore, similarity of >99% was found between HEV sequences of human and swine origins. These results indicate that consumption of some pork products, such as raw liver, is a major source of exposure for autochthonous HEV infection. [5]
As hepatitis E concentrates in the liver in both pigs and humans, swine livers were the natural place to test for hepatitis E presence, and probably the riskiest part of the pig to eat.
Further evidence that hepatitis E in pigs can infect humans was found in another French study. The researchers reasoned that sausage made from pig liver would be a likely vector for hepatitis E transmission to humans, especially a form of smoked pig liver sausage traditionally eaten raw – figatellu. Their findings:
Acute or recent HEV infection, defined by detection of anti-HEV immunoglobulin M antibodies and/or HEV RNA, was observed in 7 of 13 individuals who ate raw figatellu and 0 of 5 individuals who did not eat raw figatellu (P=.041). Moreover, HEV RNA of genotype 3 was recovered from 7 of 12 figatelli purchased in supermarkets, and statistically significant genetic links were found between these sequences and those recovered from patients who ate raw figatellu….
Our findings strongly support the hypothesis of HEV infection through ingestion of raw figatellu. [6]
The titer of hepatitis E viruses in the supermarket sausage reached as high as a million copies per slice. [6] This data suggests that a majority of figatellu in French supermarkets carries hepatitis E virus, and that a majority of people who eat figatellu acquire hepatitis E infections.
Contact with pigs can also lead to transmission; swine workers have an elevated prevalence of antibodies to HEV in the United States. [7]
Does Cooking Inactivate the Viruses?
What level of cooking is needed to inactivate the virus?
It is difficult to prove that any particular cooking or processing method renders HEV non-infectious:
How safe are these products? The question is difficult to answer because HEV grows poorly in cell culture, and in vivo testing of viability requires nonstandard laboratory animals—nonhuman primates or pigs for genotypes 3 and 4. [7]
Since scientists don’t have the funding or facilities to see if feeding cooked, cured, or smoked pork to primates or pigs gives them hepatitis E, they have no way of verifying that cooked, cured, or smoked pork is free of HEV.
In test tube experiments, HEV was still viable and infectious after cooking for 1 hour at 56°C, the temperature of rare to medium-cooked meat. [9] About 80% of viruses were inactivated after an hour at 60°C, and an hour at 70°C probably eliminates the viruses.
The implication is that thorough cooking would destroy HEV, but that some HEV will survive in rare to medium cooked pork, with liver likely having the greatest viral titer. [9] “However, much pork is consumed that has not had even that degree of cooking.” [7]
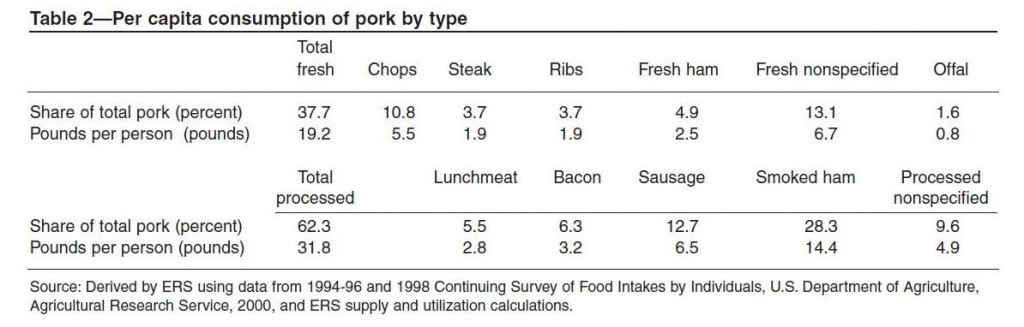
One way to reduce the risk of infection is to avoid the pig tissues that have the highest viral titers:
HEV can be found in the liver, blood, and intestinal tract, which are all consumed in one form or another and often together, such as in sausages. [7]
So: to avoid HEV infection, it’s best to avoid pork liver, intestines, or blood, or products made from them such as sausage; other cuts should be carefully rinsed of all blood and then cooked thoroughly to a temperature of at least 70°C. Simmering in near-boiling water for an hour should be sufficient.
The most dangerous pork product is likely to be sausage, which often uses pork liver meat, and traditionally uses pig intestines as the casing. It may also contain traces of pig blood. Pig blood pudding, a traditional Chinese dish, should also be avoided.
Links to Pork-Associated Liver Diseases
Hepatitis E was discovered as a cause of acute liver disease. But what about chronic diseases like alcoholic cirrhosis and liver cancer? Is there really evidence linking it to these diseases?
First, studies of organ-transplant recipients who contracted hepatitis E from their donors have shown that HEV seems to establish chronic infections in at least 58% of infected persons. [10] When anti-HEV antibodies exist, generally active viral RNA is present too. [12] So the virus is persistent.
Hepatitis B and C viruses are known causes of alcoholic liver cirrhosis. What about HEV? There have been few studies, but those that exist suggest it is likely:
- A child developed cirrhosis after a bone marrow transplant due to a swine-derived form of hepatitis E. [11]
- A Spanish study found a strong association between HEV and cirrhosis in people infected with HIV: “Liver cirrhosis was the only factor independently associated with the presence of anti-HEV, which was documented in 23% of patients with cirrhosis and 6% of patients without cirrhosis (P?=?0.002; odds ratio 5.77). HEV RNA was detected in three seropositive patients (14%), two of whom had liver cirrhosis.” [12]
- HEV seems to be a common cause of cirrhosis in Egypt. [13]
Hepatitis B and hepatitis C viruses are known causes of hepatocellular carcinoma. What about HEV? If there were few studies linking HEV to cirrhosis, there are even fewer investigating its relationship to HCC.
I did find one Chinese study showing that HEV infection greatly elevated the association of aflatoxin with HCC. (Aflatoxin, a fungal toxin that damages the liver, is a known risk factor for HCC.) [14]
Epidemiology is also suggestive. I mentioned earlier that the pork-transmitted genotypes of HEV have only recently appeared in the Americas. If HEV is responsible for alcoholic cirrhosis, hepatocellular carcinoma (HCC), or multiple sclerosis, then we should be seeing the incidence of those diseases increase. In fact, that is true for HCC:
In the U.S., incidence rates of HCC in both men and women have increased steadily during the past three decades. The reasons for this steady increase remain unknown. [15]
What About Multiple Sclerosis?
There have been no studies searching for a specific link between HEV and multiple sclerosis.
However, it may be worth reviewing what some mouse models tell us about the potential for a hepatitis virus to cause MS. MS is an infectious or autoimmune disease:
MS is felt to be most likely either due to an aberrant immune response or a pathogen, or possibly a combination of the two, and the animal models available reflect these two possible pathogeneses. [16]
Regular readers will know that I believe MS is infectious in origin. There are three animal models for MS. One of them (“experimental allergic encephalomyelitis” or EAE) involves immunizing mice with myelin or myelin proteins so that they develop antibodies to their own myelin; the other two involve infecting mice with viruses:
Two viruses, Theiler’s murine encephalomyelitis virus and murine hepatitis virus, are used to induce infectious models of the disease. [16]
The murine hepatitis virus (MHV) model is suggestive: it supports the idea that a virus that causes hepatitis may also cause MS. Some strains of MHV are neurotropic, infecting both the liver and central nervous system, and it is these that most readily produce an MS-like disease. [17]
If a hepatitis virus is causing MS in humans, we would expect MS patients to have high rates of liver disease. Indeed, there is a correlation.
MS patients are 3.7-fold more likely to have elevated ALT and 2.2-fold more likely to have elevated AST – both liver enzymes associated with liver disease. Also, elevated ALT and AST are associated with the more severe relapsing-remitting form of MS. [18]
A few perhaps insignificant links: Patients with systemic sclerosis, who are about 5-fold more likely to develop MS than others, are also at high risk for liver disease. [19] In the 1980s, doctors began observing MS patients with cases of primary biliary cirrhosis severe enough to require liver transplantation. [20]
Other Pig-Human Pathogens and MS
Pork can carry many pathogens; perhaps hepatitis E virus is not the MS-causing pathogen.
I don’t see obvious candidates however. Perhaps herpes viruses would be most likely. One of the human pathogens likely to be causal for MS is Epstein-Barr virus, also known as human herpes virus 4 (HHV-4). It causes mononucleosis but establishes persistent infections and is associated with a number of diseases, including lymphomas, MS, lupus, and rheumatoid arthritis.
Human herpes viruses may be able to establish infections in pigs. [21] And there are porcine herpes viruses that are closely related to Epstein-Barr virus. [22]
Conclusion
There is a strong association between pork consumption and liver cirrhosis mortality, liver cancer, and multiple sclerosis.
It seems likely that the association, if it is real, is mediated by a pathogen. The most likely pathogen in the case of the liver diseases is hepatitis E virus. In MS, the pathogen remains unknown, but is likely to be a virus.
Hepatitis E virus is not destroyed by casual cooking, smoking, or curing. It appears that meat must reach temperatures of 70ºC (160ºF) before viruses are inactivated; and it is possible that meat must remain at that temperature for some time, perhaps as long as an hour. Rare or medium cooked pork could contain active viruses.
Hepatitis E viruses are most abundant in liver, intestine, and blood. Pork products containing these parts, such as sausage, may be best avoided.
Meat from parts of the pig with low viral titers, such as pork ribs or pork bellies, are likely to be safe to eat as long as they are well cooked. Be sure to wash the meat of all blood before cooking, and to cook thoroughly.
Related Posts
Posts in this series:
- Pork: Did Leviticus 11:7 Have It Right?, Feb 8, 2012
- The Trouble with Pork, Part 2, Feb 15, 2012
- The Trouble With Pork, Part 3: Pathogens, Feb 22, 2012.
References
[1] Purdy MA, Khudyakov YE. Evolutionary history and population dynamics of hepatitis E virus. PLoS One. 2010 Dec 17;5(12):e14376. http://pmid.us/21203540.
[2] Purdy MA, Khudyakov YE. The molecular epidemiology of hepatitis E virus infection. Virus Res. 2011 Oct;161(1):31-9. http://pmid.us/21600939.
[3] Satou K, Nishiura H. Transmission dynamics of hepatitis E among swine: potential impact upon human infection. BMC Vet Res. 2007 May 10;3:9. http://pmid.us/17493260.
[4] Kaba M et al. Frequent transmission of hepatitis E virus among piglets in farms in Southern France. J Med Virol. 2009 Oct;81(10):1750-9. http://pmid.us/19697419.
[5] Bouquet J et al. Close similarity between sequences of hepatitis E virus recovered from humans and swine, France, 2008-2009. Emerg Infect Dis. 2011 Nov;17(11):2018-25. http://pmid.us/22099089.
[6] Colson P et al. Pig liver sausage as a source of hepatitis E virus transmission to humans. J Infect Dis. 2010 Sep 15;202(6):825-34. http://pmid.us/20695796.
[7] Purcell RH, Emerson SU. Hidden danger: the raw facts about hepatitis E virus. J Infect Dis. 2010 Sep 15;202(6):819-21. http://pmid.us/20695795.
[8] Purcell RH, Emerson SU. Hepatitis E: an emerging awareness of an old disease. J Hepatol. 2008 Mar;48(3):494-503. http://pmid.us/18192058.
[9] Emerson SU et al. Thermal stability of hepatitis E virus. J Infect Dis. 2005 Sep 1;192(5):930-3. http://pmid.us/16088844.
[10] Legrand-Abravanel F et al. Characteristics of autochthonous hepatitis E virus infection in solid-organ transplant recipients in France. J Infect Dis. 2010 Sep 15;202(6):835-44. http://pmid.us/20695798.
[11] Halac U et al. Cirrhosis due to Chronic Hepatitis E Infection in a Child Post-Bone Marrow Transplant. J Pediatr. 2012 Feb 15. [Epub ahead of print] http://pmid.us/22341950.
[12] Jardi R et al. HIV, HEV and cirrhosis: evidence of a possible link from eastern Spain. HIV Med. 2012 Jan 18. http://pmid.us/22257075.
[13] El Sayed Zaki M, Othman W. Role of hepatitis E infection in acute on chronic liver failure in Egyptian patients. Liver Int. 2011 Aug;31(7):1001-5. http://pmid.us/21733089.
[14] Tao P et al. Associated factors in modulating aflatoxin B1-albumin adduct level in three Chinese populations. Dig Dis Sci. 2005 Mar;50(3):525-32. http://pmid.us/15810636.
[15] Yuan JM et al. Synergism of alcohol, diabetes, and viral hepatitis on the risk of hepatocellular carcinoma in blacks and whites in the U.S. Cancer. 2004 Sep 1;101(5):1009-17. http://pmid.us/15329910.
[16] Pachner AR. Experimental models of multiple sclerosis. Curr Opin Neurol. 2011 Jun;24(3):291-9. http://pmid.us/21519255.
[17] Carbajal KS et al. Surgical transplantation of mouse neural stem cells into the spinal cords of mice infected with neurotropic mouse hepatitis virus. J Vis Exp. 2011 Jul 10;(53). http://pmid.us/21775959.
[18] Tremlett H et al. Liver test abnormalities in multiple sclerosis: findings from placebo-treated patients. Neurology. 2006 Oct 10;67(7):1291-3. http://pmid.us/17030771.
[19] Robinson D Jr et al. Systemic sclerosis prevalence and comorbidities in the US, 2001-2002. Curr Med Res Opin. 2008 Apr;24(4):1157-66. http://pmid.us/18430269.
[20] A patient with primary biliary cirrhosis and multiple sclerosis. Am J Med. 1992 Apr;92(4):433-6. http://pmid.us/1558090.
[21] Kim JH et al. Infection of porcine cells with human herpesviruses. Transplant Proc. 2010 Jul-Aug;42(6):2134-7. http://pmid.us/20692426.
[22] Doucette K et al. Gene expression of porcine lymphotrophic herpesvirus-1 in miniature Swine with posttransplant lymphoproliferative disorder. Transplantation. 2007 Jan 15;83(1):87-90. http://pmid.us/17220799.




















Recent Comments