Commenter qualia recently got his serum 25-hydroxy vitamin D levels tested and found a surprising result: He had doubled his vitamin D intake from 5,000 IU to 10,000 IU per day, but his 25(OH)D levels didn’t budge – they were at 61 and 62 nmol/l, equivalent to 24.4 ng/ml in American units.
24 ng/ml is well below the optimal level for healthy people of 40 ng/ml. When a healthy person supplements vitamin D, the serum 25(OH)D level usually rises linearly with dose up to about 40 ng/ml, then it rises very slowly thereafter as the body tries to keep 25(OH)D from rising by putting vitamin D into storage.
So it was natural for qualia to expect his serum 25(OH)D level to rise when he doubled his dose.
But it didn’t. The stability of his 25(OH)D levels suggests that his body has reached an equilibrium at 24 ng/ml. Instead of plateauing at 40 ng/ml with abundant vitamin D3 as a healthy person would, he is plateauing at a lower level.
Why does that happen?
Well, I don’t know. But I would like to provide qualia with a little bit of background, because this could be a clue that helps diagnose his condition and optimize treatments.
Normal vitamin D regulation strictly controls 1,25D levels
I suggested in the comment thread that qualia get his 1,25D levels measured as well as 25(OH)D.
Levels of 1,25D are not routinely measured, even in scientific studies, because they rarely vary. Blood levels of 1,25D control calcium homeostasis and are tightly regulated. In healthy people, as vitamin D intake rises from zero, serum 1,25D levels shoot up to normal levels before 25(OH)D levels reach 12 ng/ml. They then stay in a normal range no matter how high 25(OH)D levels rise. The kidney is the primary controller of blood 1,25D levels. The normal range is about 16 to 42 pg/ml (42 to 110 pmol/L).
While all human cells can convert 25(OH)D to 1,25D, most cannot release 1,25D into the blood. 25(OH)D freely crosses cell membranes and maintains the same level throughout the body; but 1,25D does not cross membranes. This allows every cell in the body to “personalize” its 1,25D levels to its own needs.
Both 25(OH)D and 1,25D are active ligands for the Vitamin D Receptor (VDR), a nuclear receptor. [1] When either 25(OH)D or 1,25D binds to the VDR, the compound is imported into the nucleus, where it combines with a vitamin A-activated Retinoid X-Receptor (RXR) to form a transcription factor which, among other roles, upregulates production of antimicrobial peptides (AMPs) that are crucial for cellular defense against intracellular pathogens.
The difference between 25(OH)D and 1,25D is that 1,25D is about fifty-fold more likely to bind to the VDR than 25(OH)D. So by converting more 25(OH)D to 1,25D, cells can upregulate their VDR activation and upregulate their immune defense against pathogens. Meanwhile, uninfected cells can keep their 1,25D levels low. Across human cells, there is a thousand-fold variation in the rate of conversion of 25(OH)D to 1,25D. [1]
In chronic infectious diseases, blood 1,25D is dysregulated
However, in people with chronic infectious diseases, 1,25D levels range all over the map, and are largely uncorrelated with 25(OH)D levels. Here is a scatter plot from a paper by Dr. Greg Blaney [2]:
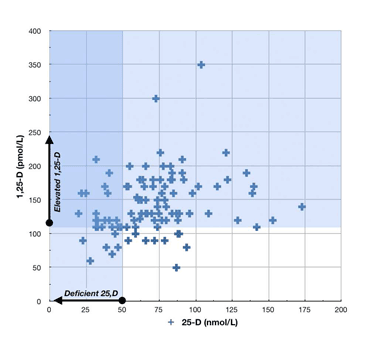
The patients in this sample were 100 chronic disease patients: 29 with fibromyalgia, 27 with chronic fatigue syndrome, 12 with post-treatment Lyme Disease, 9 with metabolic disease, 6 with osteoarthritis, 4 with irritable bowel syndrome, 4 with psoriatic arthritis, 3 with multiple sclerosis, 3 with seronegative arthritis, and 27 with other diseases.
Probably all of these diseases are caused by chronic parasitic infections.
A few things to note from this plot: (1) 25OHD levels in a lot of chronic disease patients cluster around the 61 nmol/L level that qualia has; and (2) most chronic disease patients have 1,25D levels well above the normal range, even though their 25OHD levels are mostly below the optimal level in healthy people.
What Causes 1,25D Dysregulation?
Here’s where we get into speculation. There just hasn’t been research exploring this question. Researchers are only just realizing that these diseases are infectious in origin and that vitamin-D-mediated innate immunity is critical to the intracellular immune defense.
Rather than speculate, I’m just going to mention a couple of possibilities.
First, in granulomatous diseases like sarcoidosis, it’s common to have low 25(OH)D and very high 1,25D. Granulomas are nodules where immune cells have been unable to eliminate some foreign matter and instead have built a barrier around it that walls it off from the body. Granulomas often release 1,25D to the body. Some other granulomatous diseases:
- Tuberculosis
- Leprosy
- Schistosomiasis
- Histoplasmosis
- Cryptococcosis
- Crohn’s disease
These are all infectious diseases, some of them protozoal in origin.
Second, nearly all human pathogens manufacture proteins or RNA that interfere with the innate immune response. Some are known to interfere with the VDR or with other aspects of vitamin D biology. (The HIV virus blocks the VDR entirely, one reason why it predisposes AIDS patients to infections.) It’s possible that vitamin D dysregulation is brought about by direct pathogen actions to disrupt cellular vitamin D pathways.
Conclusion
The only thing we can conclude with confidence from qualia’s vitamin D tests is that he must have a chronic infectious disease … but he knew that already.
Qualia would be best served by getting advice from an infectious disease specialist with experience in chronic diseases. Such a doctor might be able to narrow down the diagnosis. A diagnosis would help determine which antibiotics might be appropriate to help fight the infection.
Until a doctor’s diagnosis or qualia’s personal experience indicates otherwise, it’s probably prudent to continue with a reasonable intake of vitamin D and to increase iodine as quickly as possible. (Even this is not certain: the standard advice is to minimize vitamin D in granulomatous diseases.) Other infection-fighting supplements, like vitamin C, N-acetylcysteine, and glutathione are likely to be helpful also.
Finally, I always recommend that anyone with a chronic disease find a good discussion forum, like the one at http://cpnhelp.org, and try to find people with similar disease histories and learn from their experiences.
Best of luck, qualia, and please keep us posted.
References
[1] Lou YR et al. 25-Hydroxyvitamin D(3) is an agonistic vitamin D receptor ligand. J Steroid Biochem Mol Biol. 2010 Feb 15;118(3):162-70. http://pmid.us/19944755.
[2] Blaney GP et al. Vitamin D metabolites as clinical markers in autoimmune and chronic disease. Ann N Y Acad Sci. 2009 Sep;1173:384-90. http://pmid.us/19758177.











awesome post! i very much appreciate your analysis. i’m gonna do the D3 and D2, incl. calcium test next week and keep you updated 🙂
btw, my CRP was 0mg/L at the last lab a few days ago. is CRP always/also/at all elevated for chronic infections (viral, bacterial) like those you hinted at above, or just for acute invasions? wiki says for infections it would be at least 10mg/L. what do you think?
btw, meanwhile i’ve done a “self made” food allergy prick panel (heh) on my forearms and tested most stuff that lied around in my kitchen. i def. can say/confirm that i have several cross-reactions to my primary birch pollen, grass (and probably some more) allergies. the strongest “responders” were kiwi, banana, carrot, nectarines, avocado, and a bit weaker soy milk and tomato. additionally i have the related oral reactions to cashew, almonds, and walnuts. there are likely some more, but i’ve not encountered/tested all remaining foodstuffs yet. the the listed fruits and veggies create almost no oral reaction, but slight digestive and systemic reactions like lethargy, brain fog, lack of drive, dizziness, weakness, slight respiratory suppression, abdominal swelling and dull abdominal pain. the same is true for dairy and eggs, but those did not react to the prick test (except for egg-white a tiny bit). if it wasn’t for the stuck D-level, i’d say as of today i just ate all the wrong stuff for decades (incl. gluten) without knowing that it constantly created all the above mentioned symptoms incl. several nutritional deficiencies. yesterday afternoon and today i managed to avoid all the tested allergens, and felt great, even without most of my supportive supplements i usually take. then i challenged with a nectarine, and felt a slight fall-back in energy. then i challenged with canned peas+carrots, and had a clear digestive reaction for hours (swelling), and again a moderate drop in energy. right now, several hours later, i’m pretty much back to 100%. i’m also currently taking additional zinc, and B6, just FYI. zinc shows a noticeable increase in libido since a few days now, and the (activated) B6 shows a strong brain-booster effect (comparable to Phosphatidyl-Serine). also, even tho my bowel/digestion is now rather stable since like 2 weeks on gluten free, it still clearly reacts to certain foods (like above), but at least keeps stable with regards to stool quality and frequency. so the question seems to be: just some long-term complications with gluten and food allergies (incl. disbyosis and leaky gut), or do we indeed also have an infection (maybe as overall underlying cause). let’s see what the next few weeks bring.
Hi qualia,
Clearly you’ve improved a lot of things lately in diet and supplements. It will be interesting to see how many problems are relieved by another 2-3 months of healing.
Good luck.
Interesting post on the dysregulation of blood 1,25D by chronic infection.
Holick in his book, “The Vitamin D solution” claims that vitamin D from sunlight has a longer half-life than vitamin D from supplements.
Also, this study mentions that outcomes for UV radiation and Vitamin D supplements may be different.
http://www.ncbi.nlm.nih.gov/pubmed/16616326
http://www.ncbi.nlm.nih.gov/pubmed/20136910
Personally, i don’t think we can get the true benefits of sunlight in a Vitamin D pill. But I do not know the real difference in effects on 25(OH)D level between oral supplementation and sunlight exposure.
Hi Daniel,
Yes, I think everyone would agree that sunlight is better than supplements. Holick makes the point that there are five to ten other photoproducts produced by sun exposure in addition to vitamin D3. Evolution will have optimized the body for maximum benefits from exposure to sun, while there has obviously been no evolutionary optimization for vitamin D pills.
It seems virtually impossible to overdose on vitamin D from sunlight, but it’s probably quite possible from pills. With a few decades of experience from widespread D supplementation we’ll probably start hearing of overdose conditions.
That said, most of us work indoors through the day or live at northern latitudes where winter sun availability is small. The supplements are a huge improvement over vitamin D deficiency conditions. I think 2,000 IU for women and 2,500 IU for men — that’s what I take — is safe and helpful if you spend your workday indoors. More is needed in many disease conditions, and that’s where the science is really inadequate at this point. It’s not possible to give scientifically grounded advice to someone like qualia re vitamin D supplements. He has to guess.
Paul
Regarding qualia case, i think there could also be other factors that are involved in cases of vitamin D deficiency.
There are some evidences fructose could play a role.
http://jasn.asnjournals.org/cgi/content/abstract/ASN.2009080795v1
and a high fibre diet too.
http://www.ncbi.nlm.nih.gov/pubmed/6299329
Also they are some arguments that obesity plays a role in vitamin d deficiency because this vitamin is fat soluble and it is prone to getting locked up in fat cells.
Thanks for the links, Daniel. The fiber paper is actually cited in our diet book as evidence for wheat toxicity. They used wheat bran for the fiber, and it’s well known that wheat can induce rickets (Mellanby used it for that purpose in his experiments on dogs). I doubt that fiber generally depletes vitamin D – I think it is a wheat protein toxicity effect. Blueberries and sweet potatoes also have fiber, but I don’t believe anyone has ever induced rickets with them!
Of course fructose should be minimized in anyone’s diet.
alright, i’ve got some new lab test results 🙂
as you will remember, my D3 about 3 month ago was 61nmol after having re-tested the sample (or 81, if we take the original measurement), then a week ago 62nmol, and now with a different lab back to 82nmol..
in addition, this time i also measured:
1,25-Di(OH)D3 -> 214pmol/L (48-170)
Calcium -> 2.46mmol/L (2.1-2.6)
*Creatinine -> 117umol/L (59-104)
*i need to mention that i’m regularly taking creatine as a fitness supplement, and my creatinine levels always have been a bit higher, presumably because of this.
so 1,25-D3 and calcium seem to be a bit elevated, while 25-D3 has found its way back into the “sufficient” range within just 7 days.. ahem.
ok, technically this is possible, if we imagine that within those 7 days something happened with regard to the absorption rate of my D3 supplement. and indeed, i have switched my D supplement for like 2 days to a capsule/pulver form (the others are gelcaps/oil). so theoretically the D3 spike to 82nmol could be caused by this, but i’m not sure how likely this theory really is.. probably not very.
i’m also not clear what the elevated 1,25 means, or how it would look when there actually would be a problem like sarcoidosis.
on http://en.wikipedia.org/wiki/Vitamin_D i’ve found yet another hypothesis for elevated 1,25:
“Whether the toxicity of oral intake of vitamin D is due to that route being unnatural, as suggested by Fraser,[84] is not known, but there is evidence to suggest that dietary vitamin D may be carried by lipoprotein particles[91] into cells of the artery wall and atherosclerotic plaque, where it may be converted to active form by monocyte-macrophages.[92] These findings raise questions regarding the effects of vitamin D intake on atherosclerotic calcification and cardiovascular risk.”
sounds a bit scary to me. artery walls converting D3 into its active form? maybe i should just go back to 5kIU for a wile, lol. what do you think?
Hi qualia,
I would support going back to 5,000 IU. That’s slightly above the optimal dose for healthy people and shouldn’t be far wrong in an ill person. 10,000 IU is an experimental pharmacological dose and we don’t really know the effects. We don’t even have a diagnosis of your condition. I think 5,000 IU is sufficient to support immune response and safer. But you can get many varying opinions. Dr. Cannell would favor 10,000 IU or more.
In my view, since cells can convert 25OHD to the more active 1,25D if they need to, as long as you have reached a “plateau range” in 25OHD levels you’re probably pretty close to the optimum. I believe in cooperating with your body. If you body wants to be at 61 nmol/l, maybe you should let it be there, just provide enough 25OHD so it has no trouble doing so.
The elevated 1,25D is as expected and the calcium in the normal range is good. I neglected to mention in the post that chronic disease patients with elevated 1,25D usually have normal calcium.
If you’re worried about calcification of plaques from macrophage 1,25D creation, be sure to get supplemental vitamin K2.
ah, and btw, i also asked one of the biochemists that worked in the second lab how big the margin of error was for measuring D3, and she just rolled her eyes and said “you don’t really wanna know” lol. she couldn’t even tell, even after i insisted i wanna know. looks like D3 is still not that easy to measure, and standards to make measurements comparable between labs it only just very recently emerged (?). that’s a bit depressing for all those people like me who rely on such tests to determine the optimal supplementation..
you don’t read this stuff on vitaminDcouncil.org and similar sites, do we..
I think the main problem with D is that it’s so powerful, you’re measuring minute quantities (25 ng = 1,000 IU). This also lies behind some of the poisoning incidents where huge amounts of vitamin D got into pills by mistake.
In fairness to Dr. Cannell he has discussed unreliability of test readings (see http://www.vitamindcouncil.org/newsletter/2008-july.shtml, “Does It Matter What Reference Lab My Doctor Uses?”). At the time of that newsletter he was a consultant to the lab he recommended, now he has a partnership with ZRT labs and sells their kits. Don’t know if that biases his comments.
ah, indeed, thanks for the link!
i now requested some answers with regard to margin of error, technology/method used, and laws with regard to methods and inter-lab comparability of the results. curious to see what the answer will be. so far the head of the first lab has been very cooperative.
ok, i got a short answer from the head of my primary lab.
he says that there is no explicit margin of error for D measurements (which is BS of course) – just the analyzer machine that requires periodic recalibration with a given reference-sample and when this is not done as required, the machine refuses to do its job. the process used for D in this lab is “chemiluminescence” developed by Roche. also, there are no national standards for how to measure D here. just the requirement to fulfill some sort of administrative quality control, to be able to do business with insurers.
HOWEVER: when i look at this table of tests and their margin of error for example (http://is.gd/eGCZS), the possible error for D is freaking HUGE: +/- 15% (day to day). meaning, my levels of 62 and 82 could theoretically still be one and the same level, and the actual level could be 70nmol for example. pretty disappointing, considering that, say having 25ng or 32ng makes a huge difference for the body’s biochemistry. this pubmed article seems to confirm this: http://www.ncbi.nlm.nih.gov/pubmed/20397196 so, a vitamin D test basically only is useful (as of today) to identify severe states deficiencies, and/or states of hypervitaminosis. but don’t expect to be able to reliably track your progress by 10nmol steps with each measurement..
Hi qualia,
I think using amounts of D3 as well as 25OHD to judge status is prudent and probably allows you to optimize reasonably. 5,000 IU should give the body enough raw material to work with. So I wouldn’t worry too much about precision of the 25OHD number.
I bet you’ll find that when your iodine intake gets up to 50 mg/day, your 25OHD will have increased and your 1,25D decreased.
Hi
I have terrible side effects from d3 even at 1000 iu with K2 they get worse over time. Running red eyes, pains in bones and teeth, nervousness and panic to the extent of not being able to leave the house or be alone.
I have Hashimotos and I am probably low in iodine. Could you please explain your post about iodine and decrease of active vitamin D? I was tested very low for the inactive type.
regards
Dawn
Hi Dawn,
I think your immune system is starved and whenever it gets some D, it goes to work against whatever infections you have and you get the symptoms of inflammation/immune attack. I think you need to keep giving yourself D and vitamin A (liver, spinach) and manage thyorid status and work on circadian rhythm entrainment and keep working through this. Eventually your immune system will probably get the upper hand.
Serum 25OHD is not inactive and you need to get it into something near the normal range. A merely low level, say 30 ng/ml, can be a symptom of disease, but anything below 20 ng/ml indicates severe deficiency.
Thank you Paul.
I will try again when the panics wear off a bit. I have to be on steroids merely to function which of course dampen down the immune system, however maybe not at the dose I take at 30mg hc equivalent.
I have a ACTH stim test this week. I feel so fed up and lethargic, I have been housebound so long now it is no wonder I am deficient, however I started with this problem before I was housebound, the side effects. What you say sounds very probably. I am so happy you replied to me.
I am hoping to get going on your diet but my husband and family aren’t very eager to do shop for and study new eating habits and really I am stuck at what can be done for me.
I am going to buy your book and try to inspire them. Wish me luck.
You are a lovely couple
regards
Dawnx
PS what do you think of LDN? I had been considering it with all my inflammation and AI problems.
Hi Dawn,
LDN does help some people. I would recommend it. I’d also try hard to get daily sunshine on bare skin and eat liver weekly and spinach daily.
Best, Paul
Stumbled upon your site while searching for information abouit vitamin d dysreguiation and was so impressed that I had to tell you so. You gave me a much more comprehensive insight into some of vitamin D’s ecosystem that I had never imagined might be the case.
All of which prompts me to mention my vitamin D enigma that has my doctor stumped. When I retired 15 years ago as a consulting psychologist I moved from the San Francisco Bay area to the Seattle area to be close to my son. Then I got so caught up in using the computer to follow a range of interests that I seldom got out of doors — and the latitude here limits the D I could get from sunlight anyway. sunlight — and I get virtually no vitamin D from my diet since I am allergic to seafood. The upshot was that as I moved into my 80’s I was confronted with a variety of physical changes that that I now think were due to severe vitamin D deficiency. Three or four teeth just crumbled over a period of a month r so, I developed adult scoliosis, and my blood pressure (always a bit high) went out of control, hitting the 190’s and low 200’s. I refused blood pressure pills since I had previously been damaged by them, and instead began taking increasing amounts of vitamin D. When I hit 15,000 a day it began to drop, and settled at teh 150 to 175 range. Three months ago my vitamin D level was measured as part of a yearly physical exam, and when my doctor found that my NgL level was 92 he said that he had never seen one that high and asked me to cut my intake to 10,000 units for starters. I had tried to do that three times previously, and my blood pressure went back up the first two times and the third time my face began to swell. This fourth time didn’t work either, with my blood pressure going up after a few days of starting. I stuck it out for two weeks and then went back to the 15,000 IUs. But, as opposed to my three earlier tries, when blood pressure was back to my normal in a week, this time it took a six weeks before the blood pressure came down again. So the enigma that I have has to do with this weird relationship between my vitamin D “requirement” and my blood pressure.
Otherwise I feel better than fine. My Google research led me to a curcumin program a while back and that has brought back my original dark brown hair color, and recently I found that I now longer had to get up to go to the bathroom every night(as has been the case for years). And as of a week ago I found that m prostate is shrinking. More importantly, the AMD I have in both eyes is gradually reversing to the point where I no longer am a member of the enlarged print gang. So as far as I know everything is working fine and I don’t have a chronic anything!
Dear Charles, I’m glad to hear you are doing so well! Sounds like you’re another one of the cases Dr. Cannell likes to talk about whose diseases clear up with sufficiently high levels of D. It may be that everyone with a chronic disease should experiment for a month with very high levels to see what happens.
Please remember to supplement with vitamin K2 along with your D. This is very important for your blood pressure and vascular health generally, not to mention cancer and overall mortality risk. I take this one daily, I recommend you do the same: http://www.amazon.com/Life-Extension-Advanced-Complex-softgels/dp/B000MYXVV4/
Magnesium is another cofactor of D that is important, I recommend 200 mg/day magnesium citrate.
High blood pressure can have several causes, including chronic vascular infections, but in most people toxicity effects from fructose or alcohol plus omega-6 fats is a major contributor. It is not possible for me to diagnose your specific situation but I am fairly confident that high blood pressure would usually clear on our diet and supplement program. As Tolstoy might have said, all healthy people are alike, but every unhealthy person is unhealthy in his own way. It’s often easier to become healthy than to figure out why you are unhealthy.
Good luck and thanks for the compliment!
Paul
Hi Charles,
Look into the Dr Stasha Gominak on vitamin D and sleep.
Fixing the D fixes the sleep which eventually, fixes the hypertension.
You may need to take b50 vitamin for no longer than 2 months.
Increasing magnesium can helps too.
A pinch of Epsom Salts in all your drinks will provide both magnesium and sulfur.
ah, btw “upping iodine to 50mg/day” – i’m now at 20mg/day with stable, if not even recessing bromide acne. i often do a “few days ON, and few days OFF” scheme, and what i noticed is that on the “ON” days, i always pretty rapidly get dry and grayish and “old” looking skin (whole body), which you would expect to see on a heavy smoker, and on the OFF days, this reverts back also pretty fast (2-3 days) into normal, healthy looking skin again. in the “ON” phase, the skin also seems to be less sensitive to touch, and almost feels a bit numb at times. i guess the usual theory behind this probably would be “that’s the excess bromide that is mobilized” etc, but to me it looks more like excess oxidative damage(?). what do you think? is it just a whole-body bromide symptom, or can high amounts of iodine intake (IOSOL) also have “real” short term detrimental effects? thanks!
Hi qualia, That’s interesting. I don’t know what causes the grayish skin. I would be sure to drink lots of water on “ON” days. You will need much more than normal when taking the iodine, and dehydration could have something to do with it.
Charles, I’ve put up a post about your case here: http://perfecthealthdiet.com/?p=448.
I may have solved your enigma!
Cannell also talks about ‘falling vitamin D levels’ being as dangerous as low levels.
As it seems unlikely that a levels of 92 ng/mL is in any way problematic, I’m just theorizing here:
It is possible that the increase in blood pressure that qualia sees when he lowers his dose is simply the risk of which Cannell speaks. And if so, it seems reasonable that his levels might again resolve once his body grows accustomed to the new, stable level that is no longer dropping?
Hi cillakat,
I think it was Charles’s blood pressure that increased.
The issue that high doses may induce vitamin D resistance, and then insufficient VDR activation when vitamin D levels are reduced, is a real one. Presumably it could contribute to high blood pressure, just as low vitamin D can. So your idea is a good one.
But in Charles’s case, 5,000 IU didn’t work for him even before he tried high doses. So it seems his problem was not vitamin D resistance induced at the 10,000 IU dose, but rather some inability of 5,000 IU to properly stimulate the VDR.
I suggested an alternative hypothesis in my follow-up post, http://perfecthealthdiet.com/?p=448.
Best, Paul
Medical research has proven that a proper amount of vitamin d is essential to heart health, muscle growth, and bone strength at all ages. It has also been shown to be indispensable for satisfactory brain and mental function, particularly among people over the age of 50.
On the topic of Vitamin D, the importance of vitamin D in the development of cancer was further confirmed by a 2009 study by Sylvia Chistakos, Ph.D., of the UMDNJ-New Jersey Medical School. Her research, shown in The Journal of Biological Chemistry, showed that the active form of vitamin D induces the production of a protein that can stop the growth of breast cancer cells.
Hi Ryan,
Thanks for the information. The paper is here: http://www.ncbi.nlm.nih.gov/pubmed/19054766.
The difficulty with interpreting this is that at higher vitamin D intakes, intracellular 1,25D levels are inversely proportional to 25OHD levels. Since only the 1,25D inhibits breast cancer cells, taking more vitamin D to increase 25OHD might reduce 1,25D levels and cause breast cancer to proliferate.
So this doesn’t tell us the optimum vitamin D intake.
Merry Christmas!
Hi Everyone,
Vitamin D levels are optimal between 100 -125. Studies have found weight loss is optimised at that level and because it is a fat soluble vitamin you want to monitor it by lab work quarterly. More than 125 nmol/L is heading too high but most people’s are way too low and levels improve slowly. Big link between MS and vitamin D levels in Tasmania Australia with low sunshine 6 -7 times higher than in the sunshine state Queensland,
Cheers Chloe
Hi Chloe,
Yes … 100-125 nmol/L = 40-50 ng/ml which is approximately our optimal range too.
Hi Everyone,
I know this is an old post but I thought I would contribute. I too had low levels of 25OHD a few years back (20 ng/ml) and began supplementing with 3000 IU / day for three months, after which my levels were 34 ng / ml. But during that time I developed some really bad muscle twitching in my calves and other parts of my body. My doctors had initially ordered the wrong vit. D test (1,25OHD) and my levels were at 75 pg/ml. Anyway because of the twitching i stopped supplementing, and eventually the muscle twitching went away.
I also have GERD and may have CFS (just generally pretty tired), and in the last few months have developed some nerve pain in left face/hand/foot, so I’m seeing a neurologist tomorrow. I had my 25OHD levels tested again, down to 25 ng/ml. But I’m freaked out I have MS so I’ve been eating about 1.5 lbs of salmon / week and back on 1000 IU vitamin D / day, but the muscle twitches are back (although they have also come and gone over the last few years even without vit. D supplements). I’ve always wondered whether the twitching was due to low magnesium, which i’ve struggled supplementing with because of GERD. Sounds like it might also be low K2?
I’ve been voraciously sucking up health info over the last few years, but I often feel frustrated that there’s not more I can do. I was on Paleo for the GERD which has helped quite a bit, but now I’m trying to add more sweet potatoes and potatoes (rice is a no go) more in line with the PHD. I’m seeing a neuro who espouses functional medicine so I’ll ask him about the elevated 1,25OHD levels.
I have to admit Paul that I find the idea of long term antibiotics kinda scary, which I think is what you have encouraged for people with chronic infections (if that is what is causing my elevated 1,25OHD levels). If this is the correct approach to this problem I hope there will be more research and a general acceptance of the safety of this practice soon.
Thanks for all the great work you do! I’ll be ordering a copy of your book soon!
Hi Jens,
Without a diagnosis long-term antibiotics are risky.
As you’ll see in the book, we recommend a variety of dietary and lifestyle tactics in chronic disease. Antibiotics are always risky and without a diagnosis of the specific pathogen any particular antibiotic is unlikely to be effective. But sometimes it is worth trying them.
One should always supplement some K2 along with D, I think, or at least eat K2-rich foods like aged cheese.
Magnesium is quite important too, and electrolytes.
High 1,25D often leads to an excess of calcium and that can induce a relative magnesium deficiency. The high calcium / low magnesium ratio can cause muscle spasms and joint pain. That may explain your knee. Try reducing calcium intake and increasing magnesium.
Low calcium can also cause muscle twitching. (See eg http://www.ncbi.nlm.nih.gov/pubmed/17571321) You might have your serum calcium levels tested.
Thanks Paul,
I avoid all dairy and have for some time, and I’ve had serum calcium measured, which was in the normal range. I also don’t eat many nuts, and I eat quite a few greens but probably not enough on a daily basis to meet RDA’s of calcium and magnesium. I’ll pick up some K2 supplements and continue to experiment with magnesium. Anyway, good article.
And what about bone broth and fermented veg? I would think they would both help. And be sure to drink the fermented veg brine.
When you say aged cheese how do we know which cheeses are “aged” and would have good amounts of Vit K?
Greg
I don’t mean to be snarky, but you can just look that stuff up on Google. Aged cheese is not a Perfect Health Diet exclusive:
https://www.google.com/search?client=safari&rls=en&q=%22aged+cheese%22&ie=UTF-8&oe=UTF-8
Best
As far as I can tell, from a layman’s perspective and with a little googling, most cheeses are aged to some degree. But which cheese aging criteria would yield what levels of vitamin k doesn’t seem to be readily available info.
Thought someone might have spent the time trying to track that down.
Greg, It will generally say “aged over six months” or something to that effect on the label. This would imply the more traditional rennet based fermentation process has been used as opposed to the cheaper enzyme based high volume method.
HI Paul,
I was told by folks at the Vitamin D Council the following:
“Yes, if blood calcium is low, or if you’re not getting enough calcium in your diet, 1,25 can be high. If you’re not getting enough calcium in your diet, 1,25 will go high to try to search for calcium in your gut (and ultimately your bones, if there is nothing in your diet).”
Whenever I take vitamin D, despite it being low, I get bone spurs or my teeth start to feel loose. My 1,25 d though was high when it was tested. And there’s no way I get even 600 mgs of calcium, so I’m going to try to increase it or try some supplementation and will let you know if the spurs go away, along with muscle twitching and cramps.
p.s. When I take magnesium, the twitching and cramping gets worse, so I’m guessing that also indicates it’s due to low calcium.
I’d appreciate your thoughts.
Iodine appears to effect D levels.
Take a look at this.
http://curezone.com/forums/fm.asp?i=2030099#i
Fantastic post!! Did you also consider the possibility of a deficiency of NADPH, which would lead to a problem hydroxylating previtamin D to 25–D, and same for 25-D to 1,25-D? This could be due metabolism or mitochondrial issues which of course are probably caused by pathogenic microbes and/or heavy metal toxicity.
Hi Paul,
I’ve tracked the improvement or worsening of my migraines and dizziness after the use of different supplements. I’ve long suspected infection. When I increase my vitamin d levels through supplementation or exposure to sun, I always feel worse–brain fog, increased frequency of headache, nervousness, etc. I’ve replicated it several times.
It’s curious to me that this would happen. I wonder if it is the increased immune activity that is creating symptoms. Or that I have some infection like the ones you list in your blog post. If someone were to get sick with increased vitamin d should they consider getting tested for the 1,25-D?
Hi Jim,
I think your guesses are good ones. I would start by getting tested for 25OHD, 1,25D, and calcium, and see if any of them seem off. The first thing to make sure is that you are not overdosing on D.
Assuming you are achieving only normal levels of D, then it seems likely that increased immune activity (ie, a restoration to normal levels) is a likely factor. But it’s hard to say, since D has such wide-ranging effects, and is coupled to many other nutrients including vitamin A. For example, vitamin D could aggravate a vitamin A deficiency which in turn would aggravate hypothyroidism and circadian rhythm disruption. So be sure to follow all the other PHD nutrition and lifestyle advice.
Best, Paul